 |
 |
While many optometrists may think that anterior diseases such as keratoconus and corneal scarring are the chief indicators for corneal transplantation, endothelial decompensation is actually the primary indication. This broad condition, which includes Fuchs’ endothelial corneal dystrophy (FECD), pseudophakic and aphakic corneal edema and failed corneal transplantation, accounted for 60% of the 51,000 corneal grafts performed in the US in 2019.1 In all of these cases, surgery was the only option available to successfully restore vision.
Modern endothelial transplant surgeries, such as Descemet’s membrane endothelial keratoplasty (DMEK), Descemet’s stripping automated endothelial keratoplasty (DSAEK) and Descemet’s stripping only (DSO)—also known as descemetorhexis without endothelial keratoplasty (DWEK)—are associated with a substantially improved course, recovery and complication rate compared with penetrating keratoplasty. However, the post-op period can be complicated by detachment of the transplant requiring rebubbling, failure of the transplant and patient positioning restrictions, which are all problems that come up rather frequently, even among the most experienced surgeons.
Given the limitations of surgery and recovery, a medical approach to managing corneal edema would be beneficial to patients, optometrists and cornea specialists alike. Interestingly, we may already have access to something of a similar nature.
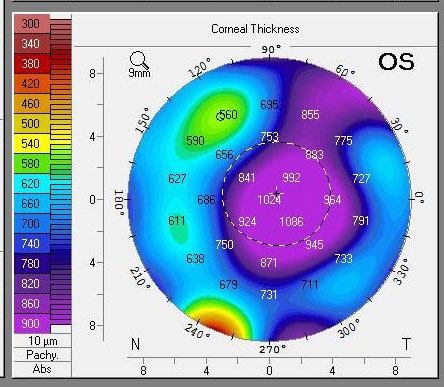 |
| Pachymetry following a DSO supported by Rhopressa shows profound central edema. Click image to enlarge. |
Background
Rho-associated kinase (ROCK) inhibitors were recently introduced for the management of glaucoma. In 2017, Rhopressa (netarsudil, Aerie) became the first widely available ROCK-inhibiting agent on the market.2 This family of molecules produces an IOP-lowering effect via its impact on the cellular cytoskeletons and actin filaments, which creates an anatomical shift in the architecture of the trabecular meshwork. This shift reduces resistance to outflow, thereby lowering IOP.
Interestingly, but perhaps unsurprisingly, the potential of ROCK inhibitors seems to extend beyond the management of glaucoma. The ROCK pathway was identified as a potential target of intravitreal therapy in diabetic retinopathy as far back as 2010, and trials are ongoing.3 It is possibly within corneal disease, however, that these molecules may alter the management paradigm of disease the most dramatically.
In 2013, a group out of Japan found that the ROCK inhibitor Y-27632 was able to restore corneal clarity and endothelial cell density (ECD) after a short course of treatment in a primate model following transcorneal freezing (to kill endothelial cells). The treated animals had twice the ECD of controls by six weeks post-injury.4
The same team published a case report shortly after, in which a patient with FECD treated with the same ROCK inhibitor went from a pre-treatment central corneal thickness (CCT) of 757μm and peripheral ECD of 757c/mm2 to a CCT of 568μm and ECC of 1,549c/mm2 post-treatment. The researchers also pointed out that the structure of post-treatment corneas did not exhibit the polymegethism and pleomorphism often seen on specular microscopy after endothelial injury, and instead had a shape and size consistent with a healthy endothelium. They speculated that in addition to impacting cellular adhesion and migration of the endothelium, these results also suggest that treatment with this particular ROCK inhibitor may increase proliferation of the endothelium.5 As the endothelium is generally considered non-miotic and arrested in the cell cycle, the ability to produce short bouts of endothelial replication and migration has enormous potential in the management of corneal pathology.
Since that time, ROCK inhibitors have been used as adjunctive therapy for DSO, a procedure for patients with localized, central FECD, during which the central endothelium is removed and not replaced with a transplant. The goal is to remove the light-scattering influence of guttata and allow the normal endothelium to fill in this gap. The addition of ROCK inhibitors is thought to facilitate a more rapid clearing of postoperative edema and potentially improve ECD.
Recently, the Massachusetts Eye and Ear Infirmary published a cases series on patients who received Rhopressa in an attempt to facilitate the clearing of corneal edema that would likely otherwise lead to transplantation. A patient with iridocorneal endothelial (ICE) syndrome-induced corneal edema, a patient with early corneal graft failure resulting in corneal edema and a patient with chronic corneal graft failure resulting in corneal edema all responded to once-daily dosing with netarsudil. This effect was measureable by one month and was maintained even after discontinuation of the drop (the ICE patient continued with the regimen for glaucoma control). Of note, the series also contains one case of pseudophakic bullous keratopathy that did not respond to therapy.6
The study points out that it is difficult to predict who will respond to netarsudil and won’t. It notes that, regardless of ultimate response, all cases of corneal edema treated with netarsudil will develop prominent honeycomb/macrocystic corneal edema, which will either clear as corneal deturgescence takes place with the drop or remain in unresponsive cases and clear on discontinuation of netarsudil. This series is unique in that, with the exception of the Japanese research group’s findings in 2013, there have been no further reports of the purely medical treatment of corneal edema with ROCK inhibitors.
The variety of pathologies treated in the series is important, as it suggests the exact source of endothelial dysfunction is unimportant when it comes to efficacy. That’s not to say you should treat herpes simplex virus endotheliitis or acute corneal graft rejection with netarsudil alone. You still need to treat underlying inflammatory or infectious pathology accordingly. It does appear, however, that some cases of corneal decompensation may respond to netarsudil, regardless of the etiology.
Finally, given the impact of guttata on corneal optics, a patient with FECD may still have reduced vision due to guttata alone even if they successfully respond to netarsudil therapy. Consider, though, that the FECD patient from the Japanese case report described earlier had VA of 20/20 following treatment with the alternate ROCK inhibitor Y-27632.4
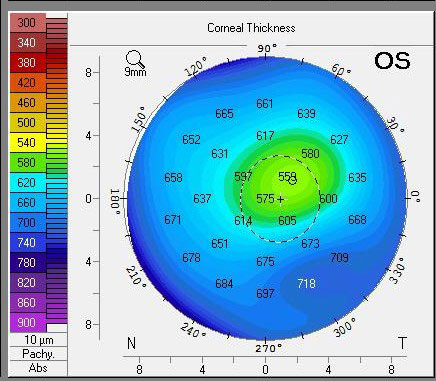 |
| Pachymetry of the same eye three months later shows dramatic clearing of central edema with continued use of Rhopressa. It’s unclear whether the DSO, Rhopressa or a combination had the most profound impact. Click image to enlarge. |
Conclusion
The small case series out of Massachusetts lends support to the use of netarsudil in the management of non-clearing corneal edema. While a randomized, double-blind interventional study on the corneal effects of netarsudil would be more telling, there is no such research currently available. Although this would take an off-label approach—and so the medication would likely not be covered by insurance—beyond cost, there appears to be little harm in a one-month trial of once-daily netarsudil. Common side effects of the medicine used in this capacity include redness, verticillata and honeycomb/macrocystic edema, but all should clear with cessation of the drug. While medical risks associated with this therapy are slight, the potential benefit of avoiding or delaying a corneal transplant may be profound.
In my (Dr. Bronner) opinion, the pros of using Rhopressa in non-clearing corneal edema tend to outweigh the cons. In the absense of research to the contrary, I fully intend to use this approach prior to considering DSAEK and DMEK. If you decide to do the same, again, understand that it is off-label, educate the patient on the resultant expected cost of therapy, take pre- and post-treatment pachymetries at similar times of the day and, if using ultrasound pachymetry, measure the same part of the cornea to ensure you are comparing equivalent measurements.
| 1. Eye Banking Statistical Report, Eye Bank Association of America. restoresight.org/what-we-do/publications/statistical-report/. Published 2019. Accessed January 19, 2021. 2. Moshifar M, Parker L, Birdsong OC, et al. Use of rho kinase inhibitors in ophthalmology: a review of the literature. Med Hypothesis Discov Innov Ophthalmol. 2018;7(3):101-11. 3. Arita R, Hata Y, Ishibashi T. ROCK as a therapeutic target of diabetic retinopathy. J Ophthalmol. 2010;2010:175163. 4. Okumura N, Koizumi N, Kay EP, et al. The ROCK inhibitor eye drop accelerates corneal endothelial wound healing. Invest Ophthalmol Vis Sci. 2013;54:2493-502. 5. Koizumi N, Okumura N, Ueno M, et al. Rho-associated kinase inhibitor eye drop treatment as a possible medical treatment for Fuchs corneal dystrophy. Cornea. 2013;32(8):1167-70. 6. Davies E. Case series: novel utilization of rho-kinase inhibitor for the treatment of corneal edema. Cornea. 2021;40(1):116-20. |


